AbstractDoxorubicin (DOX) is a widely used chemotherapy drug for various cancers and it is known to induce cognitive impairment. The aim of this study was to investigate the effect of treadmill exercise on chemotherapy-induced memory impairment. We assessed whether DOX affects inflammation, mitochondrial Ca2+ retention capacity, and Wnt/β-catenin signaling. Male Sprague-Dawley rats were divided into control group, exercise group, DOX-injection group, and DOX-injection and exercise group. To create a DOX-induced memory impairment model, animals were injected intraperitoneally with DOX (2 mg/kg) dissolved in saline solution once a week for 4 weeks. Treadmill exercise was performed once a day, 5 days a week, for 8 consecutive weeks. Short-term memory was determined using the step-down avoidance test. Western blot was performed for the proinflammatory cytokines, Wnt/β-catenin signaling, brain-derived neurotrophic factor (BDNF), tropomyosin receptor kinase B (TrkB) in the hippocampus. Mitochondrial Ca2+ retention capacity in the hippocampus was also measured. DOX-injection rats showed deterioration of short-term memory along with decreased expression of BDNF and TrkB in the hippocampus. Levels of the proinflammatory cytokines, tumor necrosis factor-α and interleukin-6, were increased in the DOX-injection rats. Wnt/β-catenin signaling was activated and mitochondrial Ca2+ retention capacity was decreased in the DOX-injection rats. However, treadmill exercise alleviated short-term memory impairment, decreased proinflammatory cytokines, increased BDNF and TrkB expression, and enhanced mitochondrial Ca2+ retention capacity. Treadmill exercise restorated Wnt/β-catenin signaling pathway. This study demonstrated that treadmill exercise can be used for patients undergoing chemotherapy with DOX.
INTRODUCTIONDoxorubicin (DOX) is a widely used chemotherapy drug for a variety of cancers and has demonstrated efficacy in solid tumors. However, DOX can cause cognitive decline and cardiovascular toxicity due to toxicity on brain, heart, and muscles. Cancer chemotherapy is known to cause cognitive decline and depression (Vitali et al., 2017). Chemobrain refers to a condition that causes thinking and memory problems during or after chemotherapy (Vardy and Tannock, 2007).
The hippocampus, a complex brain structure, connects with other brain regions and plays an important role in memory consolidating and memory maintenance (Nadel and Peterson, 2013). Chemotherapy causes cognitive impairment by reducing hippocampal volume and inhibiting nerve conduction (Brown et al., 2021; Christie et al., 2012). DOX itself does not cause direct damage to the brain, but indirectly causes brain toxicity (Andryszak et al., 2018). According to Yang et al. (2015), DOX forms tumor necrosis factor (TNF)-α in the brain, generates reactive oxygen species and anion radicals. Increased inflammatory substances and neurotoxicity in the brain are considered major factors contributing to decreased cognitive function and increased depression-related symptoms. Oxidative damage increases TNF-α production, which contributes to mitochondrial dysfunction and neuronal cell death (Keeney et al., 2015).
Wnt/β-catenin signaling is involved in learning and memory by regulating synaptic function and neuroplasticity (Rosso and Inestrosa, 2013). Wnt/β-catenin signaling is known to affect cognitive function by regulating neurotransmitters by acting on synaptic damage, neurogenesis, and apoptosis. DOX injection caused mitochondrial dysfunction and promotes apoptosis along with an increase of oxidative stress (Kitamura et al., 2015). Neuroplasticity is especially influenced by brain-derived neurotrophic factor (BNDF). Decreased BNDF level is associated with memory impairment. BDNF and its receptor, tropomyosin receptor kinase B (TrkB), are known to play a role in regulating cognitive function and depression (Gómez-Pinilla et al., 2002).
Regular exercise increases BDNF and synaptogenesis and improves neuronal function (van Praag et al., 1999). Exercise increases cerebral metabolism and nerve cell activation and restores cognitive function (Parker et al., 2017). The aim of this study was to investigate the impact of exercise on chemotherapy-induced memory impairment. We assessed whether DOX affects inflammation, mitochondrial Ca2+ retention capacity, and Wnt/β-catenin signaling.
MATERIALS AND METHODSExperiment animalsMale Sprague-Dawley rats, 8 weeks old and 200 g weighing, were used in this experiment. Experimental procedures followed the regulations of the National Institutes of Health and the guidelines of the Korean Academy of Medical Sciences. This study was approved by the Institutional Animal Care and Use Committee of Kyung Hee University (Seoul, Korea) (KHUASP [SE]-20-634). Rats were randomly divided into four groups (n=10 in each group): control group, exercise group, DOX-injection group, and DOX-injection and exercise group.
DOX-induced memory impairmentWe obtained DOX from Tokyo Chemical Industry (Tokyo, Japan). As a model of DOX-induced memory impairment, DOX (2 mg/kg) dissolved in saline was injected intraperitoneally once a week for 4 weeks as previously described (Park et al., 2019).
Exercise protocolAs described previously (Park et al., 2019), after a week, rats in the treadmill exercise groups were allowed to run on the treadmill for 30 min once a day, 5 days a week, for 8 consecutive weeks. The exercise load consisted of a speed of 3 m/min for the first 5 min and a speed of 8 m/min for the last 25 min.
Step-down avoidance testA step-down avoidance test was performed to evaluate short-term memory, as described previously (Shin et al., 2019). Animals were placed on a 7×25-cm platform with a height of 2.5 cm and left on the platform for 3 min. The platform faced a 42×25-cm grid consisting of parallel steel bars of 0.1 cm aperture spaced 1 cm apart. In training sessions, rats received a 1 mA scrambled foot shock for 2 sec immediately after dismounting. After 2 hr of training, the latency (sec) of each group was measured. Latency exceeding 300 sec was counted as 300 sec.
Concentrations of proinflammatory cytokinesConcentrations of proinflammatory cytokines, such as TNF-α and IL-6, were detected by enzyme-linked immunoassay using an enzyme immunoassay kit (Abcam, Cambridge, UK) as previously described (Kim et al., 2021).
Western blottingHippocampal tissue was homogenized on ice using a homogenizer and lysed in lysis buffer, as previously described (Shin et al., 2019). Protein content was measured using the Bio-Rad colorimetric protein assay kit (Bio-Rad, Hercules, CA, USA). Protein was separated on an SDS-polyacrylamide gel, transferred to a nitrocellulose membrane, and incubated with mouse β-actin, BDNF, TrkB, Wnt, and β-catenin antibody. Horseradish peroxidase-conjugated anti-mouse for β-actin, BDNF, TrkB, Wnt, and β-catenin was used as the secondary antibody.
Mitochondria Ca2+ retention capacityTo measure the patency of the permeability transition pore (PTP), mitochondrial Ca2+ retention capacity was determined following a previously described method (Park et al., 2019). Mitochondrial Ca2+ retention capacity was assessed using a Spex Fluormax 4 spectrofluorometer. We measured overlapping traces of Ca2+-induced fluorescence changes by calcium green-5N by continuously monitoring ΔF/min at 37°C during state 4 conditions. After determining background ΔF, the reaction was initiated by adding Ca2+ pulses (30 μM) with innervation and emission wavelength at 506 and 532 nm. Mitochondrial Ca2+ retention capacity prior to PTP opening (Ca2+ release) was expressed as picomole/mg tissue weight.
Data analysisTo confirm the expressions of BDNF, TrkB, Wnt, and β-catenin, the detected bands were counted by densitometry. IBM SPSS Statistics ver. 26.0 (IBM Co., Armonk, NY, USA) was used for statistical analysis. Data were evaluated using one-way analysis of variance followed by Tukey post hoc test. All values are expressed as mean±standard error of the mean. P-value less than 0.05 was considered significant.
RESULTSShort-term memory
Fig. 1 shows the results of step-down avoidance test. Short-term memory was deteriorated in the DOX-injected rats (P<0.05). However, treadmill exercise alleviated short-term memory impairment in the DOX-injected rats (P<0.05).
TNF-α and IL-6 concentrations
Fig. 2 shows the concentrations of TNF-α and IL-6 in in the hippocampus. Concentrations of TNF-α and IL-6 were enhanced in the DOX-injected rats (P<0.05). However, treadmill exercise suppressed concentrations of TNF-α and IL-5 in the DOX-injected rats (P<0.05).
Wnt and β-catenin expressionsExpressions of β-catenin and Wnt in the hippocampus were analyzed (Fig. 3). Expressions of phosphorylated β-catenin and Wnt were increased in the DOX-injected rats (P<0.05). However, treadmill exercise decreased phosphorylated β-catenin and Wnt expressions in the DOX-injected rats (P<0.05).
BDNF and TrkB expressionsExpressions of BDNF and TrkB in the hippocampus were analyzed (Fig. 4). Expressions of BDNF and TrkB were decreased in the DOX-injected rats (P<0.05). However, treadmill exercise increased expressions of BDNF and TrkB in the DOX-injected rats (P<0.05).
Mitochondrial Ca2+ retention capactity
Fig. 5 confirms the results of mitochondrial Ca2+ retention capacity in the hippocampus. Mitochondrial Ca2+ retention capacity was decreased in the DOX-injected rats (P<0.05). However, treadmill exercise increased mitochondrial Ca2+ retention capacity in the DOX-injected rats (P<0.05).
DISCUSSIONDOX causes short-term memory decline and cognitive impairment (Eide and Feng, 2020). In this study, short-term memory was deteriorated in the DOX-administrated rats, similar as previous study (Alharbi et al., 2020). Treadmill exercise restored reduced short-term memory in the DOX-administrated rats.
DOX causes neuroinflammation, oxidative stress, endoplasmic reticulum stress, and behavioral changes (Liao et al., 2018). DOX injection increased TNF-α and IL-6 expressions, resulting in cognitive decline (El-Agamy et al., 2018; Ren et al., 2017). Meanwhile, aerobic exercise reduced concentrations of inflammatory cytokines (Dvořáková-Lorenzová et al., 2006). Exercise suppressed hippocampal cytokine levels and enhanced antioxidant defense mechanism (Jahangiri et al., 2019). In this study, DOX injection enhanced TNF-α and IL-6 expressions in the hippocampus, in contrast, treadmill exercise suppressed TNF-α and IL-6 expressions in the DOX-injected rats.
The Wnt/β-catenin signaling pathway is a key mechanism and essential pathway for maintaining homeostasis through cell proliferation, development, migration, apoptosis, and genetic stability (Kahn, 2014; Nusse and Clevers, 2017). Wnt/β-catenin signaling acts on synaptic damage, neurogenesis, and apoptosis (Kahn, 2014; Zhang et al., 2011). In this study, DOX injection increased the expressions of Wnt and phosphorylated β-catenin, which is an overactivation of Wnt/β-catenin signaling. Meanwhile, treadmill exercise decreased expressions of Wnt and phosphorylated β-catenin, indicating restoration of Wnt/β-catenin signaling.
BDNF improves cognitive function in patients receiving anticancer drugs (Colucci-D’Amato et al., 2020). BDNF is an effective regulator of the nervous, endocrine, and immune system and is associated with nerve repair and survival, dendritic and axonal growth, and organ strengthening. Moreover, a positive correlation has been reported between BDNF level and cognitive function improvement in cancer patients (Christie et al., 2012). BDNF and TrkB expressions were decreased in DOX-injected rats, but BDNF and TrkB expressions were increased during treadmill exercise in DOX-injected rats.
DOX affects cellular oxidative stress, specifically causing mitochondrial dysfunction and neuronal mitochondrial toxicity (Andryszak et al., 2018; Yang et al., 2015). In this study, the DOX-injected rats showed mitochondrial calcium dysfunction, contributing to reduced mitochondrial Ca2+ retention capacity. In contrast, treadmill exercise improved mitochondrial Ca2+ retention capacity in DOX-injected rats. This suggests that DOX is a major cause of memory impairment through calcium dysregulation.
This study demonstrated that DOX-induced memory impairment was caused by brain inflammation and mitochondrial dysfunction through overactivation of Wnt/β-catenin signaling. Treadmill exercise ameliorated memory impairment by suppression of inflammation and restoration of mitochondrial function through normalization of Wnt/β-catenin signaling. Therefore, the present study demonstrated treadmill exercise can be used to ameliorate memory impairment in patients undergoing chemotherapy with DOX.
ACKNOWLEDGMENTSThis work was supported by the Ministry of Education of the Republic of Korea and the National Research Foundation of Korea (NRF-2017S1A5B5A07060388).
REFERENCESAlharbi I, Alharbi H, Almogbel Y, Alalwan A, Alhowail A. Effect of metformin on doxorubicin-induced memory dysfunction. Brain Sci. 2020;10:152
Andryszak P, Wiłkość M, Żurawski B, Izdebski P. Verbal memory in breast cancer patients treated with chemotherapy with doxorubicin and cyclophosphamide. Eur J Cancer Care (Engl). 2018;Jan. 27:1. 10.1111/ecc.12749. [Epub].
Brown T, McElroy T, Simmons P, Walters H, Ntagwabira F, Wang J, Byrum SD, Allen AR. Cognitive impairment resulting from treatment with docetaxel, doxorubicin, and cyclophosphamide. Brain Res. 2021;1760:147397
Christie LA, Acharya MM, Parihar VK, Nguyen A, Martirosian V, Limoli CL. Impaired cognitive function and hippocampal neurogenesis following cancer chemotherapy. Clin Cancer Res. 2012;18:1954–1965.
Colucci-D’Amato L, Speranza L, Volpicelli F. Neurotrophic factor BDNF, physiological functions and therapeutic potential in depression, neurodegeneration and brain cancer. Int J Mol Sci. 2020;21:7777
Dvořáková-Lorenzová A, Suchánek P, Havel PJ, Stávek P, Karasová L, Valenta Z, Tintĕra J, Poledne R. The decrease in C-reactive protein concentration after diet and physical activity induced weight reduction is associated with changes in plasma lipids, but not interleukin-6 or adiponectin. Metabolism. 2006;55:359–365.
Eide S, Feng ZP. Doxorubicin chemotherapy-induced “chemo-brain”: meta-analysis. Eur J Pharmacol. 2020;881:173078
El-Agamy SE, Abdel-Aziz AK, Wahdan S, Esmat A, Azab SS. Astaxanthin ameliorates doxorubicin-induced cognitive impairment (chemobrain) in experimental rat model: impact on oxidative, inflammatory, and apoptotic machineries. Mol Neurobiol. 2018;55:5727–5740.
Gómez-Pinilla F, Ying Z, Roy RR, Molteni R, Edgerton VR. Voluntary exercise induces a BDNF-mediated mechanism that promotes neuroplasticity. J Neurophysiol. 2002;88:2187–2195.
Jahangiri Z, Gholamnezhad Z, Hosseini M. The effects of exercise on hippocampal inflammatory cytokine levels, brain oxidative stress markers and memory impairments induced by lipopolysaccharide in rats. Metab Brain Dis. 2019;34:1157–1169.
Keeney JT, Miriyala S, Noel T, Moscow JA, Clair DKS, Butterfield DA. Superoxide induces protein oxidation in plasma and TNF-α elevation in macrophage culture: insights into mechanisms of neurotoxicity following doxorubicin chemotherapy. Cancer Lett. 2015;367:157–161.
Kim SH, Ko IG, Jin JJ, Hwang L, Kim BK, Baek SS. Study on the pathogenesis of liver injury caused by alcohol and drugs. J Exerc Rehabil. 2021;17:319–323.
Kitamura Y, Hattori S, Yoneda S, Watanabe S, Kanemoto E, Sugimoto M, Kawai T, Machida A, Kanzaki H, Miyazaki I, Asanuma M, Sendo T. Doxorubicin and cyclophosphamide treatment produces anxiety-like behavior and spatial cognition impairment in rats: possible involvement of hippocampal neurogenesis via brain-derived neurotrophic factor and cyclin D1 regulation. Behav Brain Res. 2015;292:184–193.
Liao D, Xiang D, Dang R, Xu P, Wang J, Han W, Fu Y, Yao D, Cao L, Jiang P. Neuroprotective effects of dl-3-n-butylphthalide against doxorubicin-induced neuroinflammation, oxidative stress, endoplasmic reticulum stress, and behavioral changes. Oxid Med Cell Longev. 2018;2018:9125601
Nadel L, Peterson MA. The hippocampus: part of an interactive posterior representational system spanning perceptual and memorial systems. J Exp Psychol Gen. 2013;142:1242–1254.
Nusse R, Clevers H. Wnt/β-catenin signaling, disease, and emerging therapeutic modalities. Cell. 2017;169:985–999.
Park SS, Park HS, Jeong H, Kwak HB, No MH, Heo JW, Yoo SZ, Kim TW. Treadmill exercise ameliorates chemotherapy-induced muscle weakness and central fatigue by enhancing mitochondrial function and inhibiting apoptosis. Int Neurourol J. 2019;23:Suppl 1. S32–S39.
Parker L, Shaw CS, Stepto NK, Levinger I. Exercise and glycemic control: focus on redox homeostasis and redox-sensitive protein signaling. Front Endocrinol (Lausanne). 2017;8:87
Ren X, St Clair DK, Butterfield DA. Dysregulation of cytokine mediated chemotherapy induced cognitive impairment. Pharmacol Res. 2017;117:267–273.
Rosso SB, Inestrosa NC. WNT signaling in neuronal maturation and synaptogenesis. Front Cell Neurosci. 2013;7:103
Shin MS, Kim TW, Park SS, Ko IG, Kim CJ, Kim M, Roh SY, Kim KT, Kim KH. Long-term surgical and chemical castration deteriorates memory function through downregulation of PKA/CREB/BDNF and c-Raf/MEK/ERK pathways in hippocampus. Int Neurourol J. 2019;23:116–124.
van Praag H, Christie BR, Sejnowski TJ, Gage FH. Running enhances neurogenesis, learning, and long-term potentiation in mice. Proc Natl Acad Sci U S A. 1999;96:13427–13431.
Vardy J, Tannock I. Cognitive function after chemotherapy in adults with solid tumours. Crit Rev Oncol Hematol. 2007;63:183–202.
Vitali M, Ripamonti CI, Roila F, Proto C, Signorelli D, Imbimbo M, Corrao G, Brissa A, Rosaria G, de Braud F, Garassino MC, Lo Russo G. Cognitive impairment and chemotherapy: a brief overview. Crit Rev Oncol Hematol. 2017;118:7–14.
Fig. 1The effects of treadmill exercise on short-term memory in step-down avoidance test. Data are presented as the mean±standard error of the mean. *P<0.05 compared to the control group. #P<0.05 compared to the doxorubicin (DOX)-injection group. A, control group; B, exercise group; C, DOX-injection group; D, DOX-injection and exercise group. 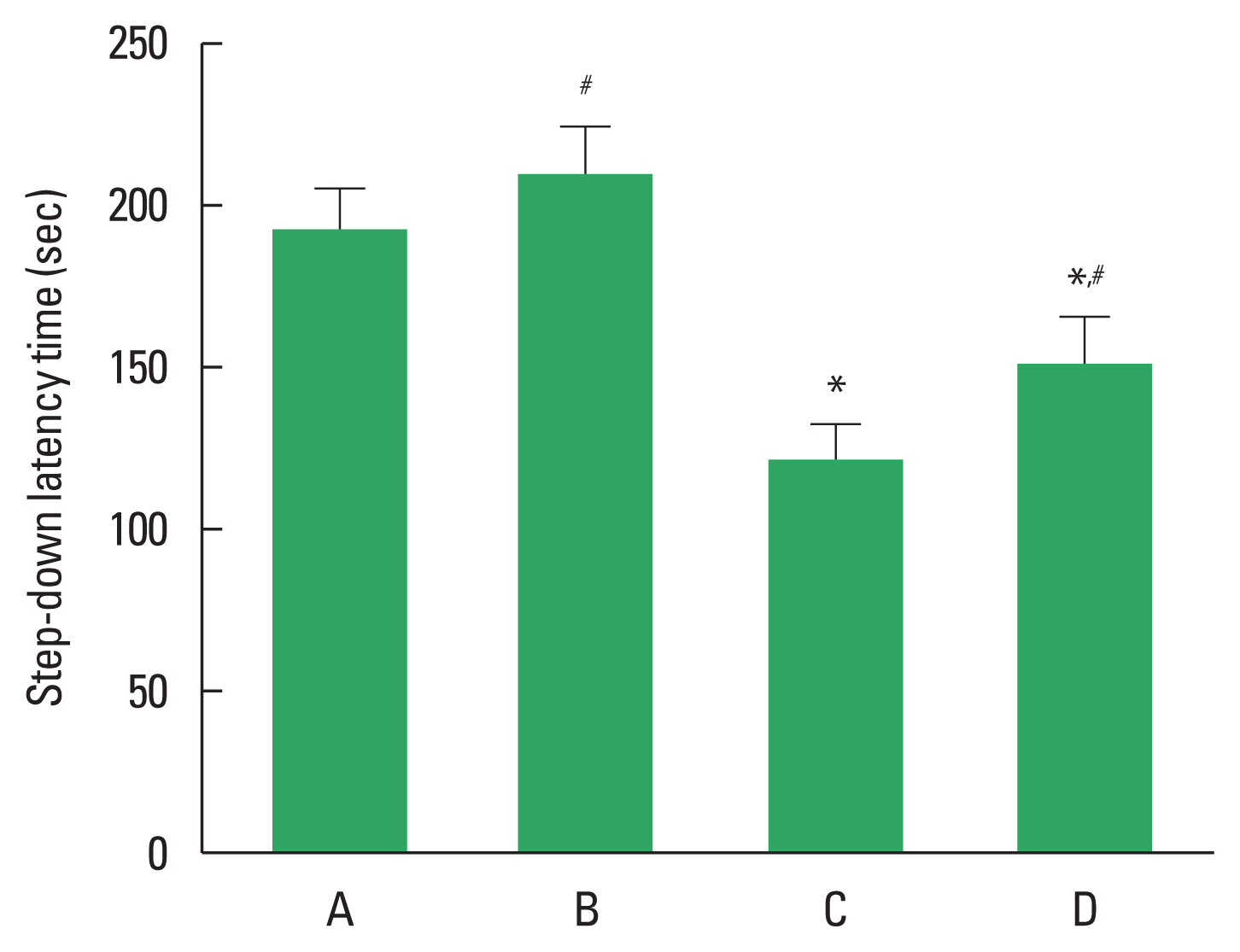
Fig. 2The effects of treadmill exercise on concentrations of tumor necrosis factor (TNF)-α (left panel), interleukin (IL)-6 (right panel) in the hippocampus. Data are presented as the mean±standard error of the mean. *P<0.05 compared to the control group. #P<0.05 compared to the doxorubicin (DOX)-injection group. A, control group; B, exercise group; C, DOX-injection group; D, DOX-injection and exercise group. 
Fig. 3The effects of treadmill exercise on β-catenin and Wnt protein expressions in the hippocampus. Upper panel: representative expressions of β-catenin and Wnt. Left lower panel: ratio of phosphorylated (p) β-catenin to β-catenin expression. Right lower panel: ratio of Wnt expression. Data are presented as the mean± standard error of the mean. *P<0.05 compared to the control group. #P<0.05 compared to the doxorubicin (DOX)-injection group. A, control group; B, exercise group; C, DOX-injection group; D, DOX-injection and exercise group. 
Fig. 4The effects of treadmill exercise on brain-derived neurotrophic factor (BDNF) and tropomyosin receptor kinase B (TrkB) expressions in the hippocampus. Upper panel: representative expressions of BDNF and TrkB. Left lower panel: relative BDNF expression. Right lower panel: relative TrkB expression. Data are presented as the mean±standard error of the mean. *P<0.05 compared to the control group. #P<0.05 compared to the doxorubicin (DOX)-injection group. A, control group; B, exercise group; C, DOX-injection group; D, DOX-injection and exercise group. 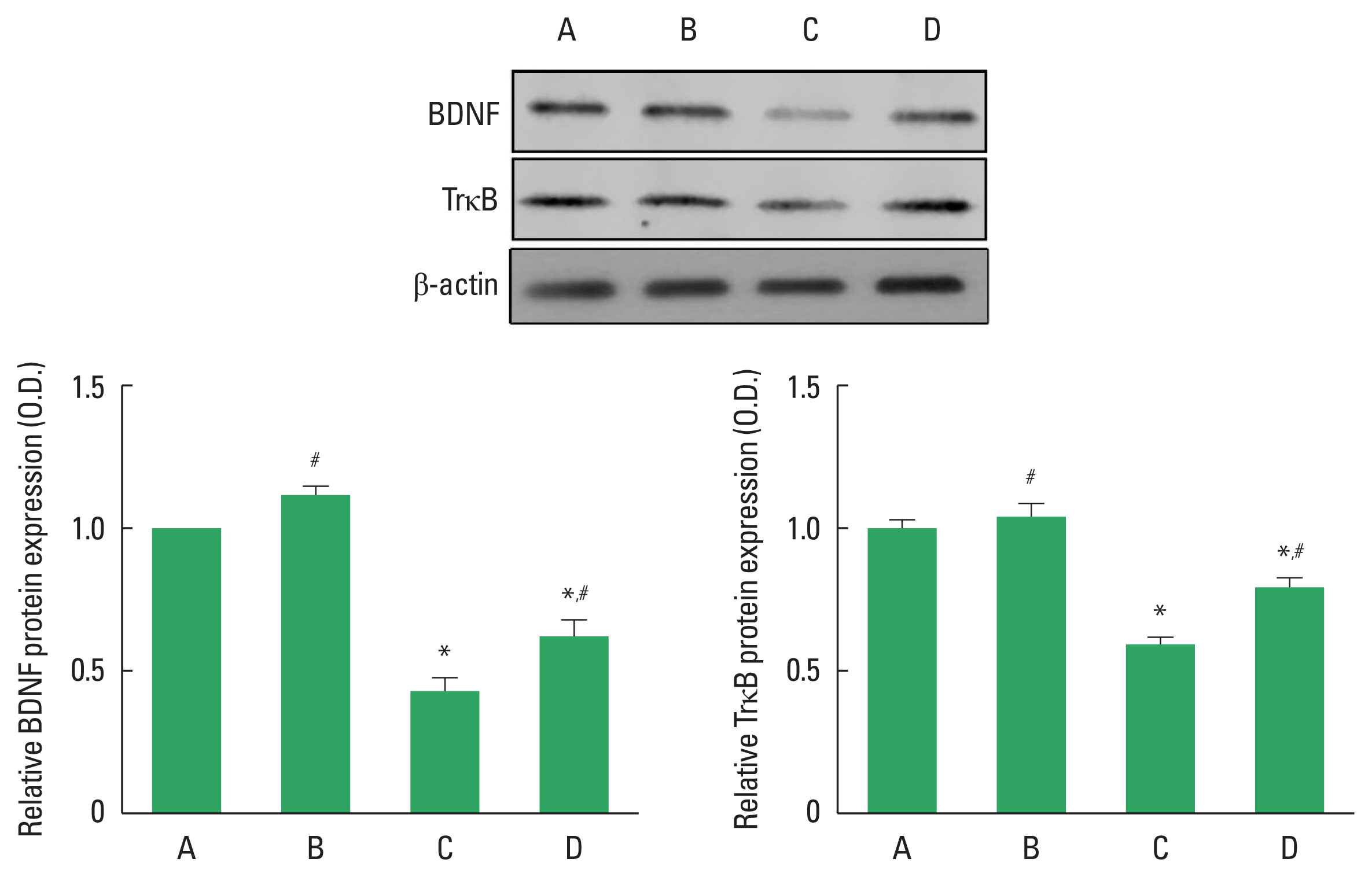
Fig. 5The effects of treadmill exercise on mitochondrial Ca2+ retention capacity in the hippocampus. Data are presented as the mean±standard error of the mean. *P<0.05 compared to the control group. #P<0.05 compared to the doxorubicin (DOX)-injection group. A, control group; B, exercise group; C, DOX-injection group; D, DOX-injection and exercise group. 
|
|
|||||||||||||||||||||||||||||||||||||